120 matters were approved!The results of the Hubei Provincial Drug Administration reported this week's approval results.
Author:Changjiang Daily Time:2022.07.04
1. Drug production license
(1) Drug production license issuance and change
1. Wuhan Zhengtong Pharmaceutical Co., Ltd.'s "Drug Production License" production address and production scope of production (trusteter);
2. Hubei Hongyuan Pharmaceutical Technology Co., Ltd.'s "Drug Production License" production address and production range and production scope of production (commissioner);
3. Changda Pharmaceutical (China) Co., Ltd.'s "Drug Production License" production address and production scope of production (commissioner);
4. Yichang Renfang Fushain Co., Ltd. changes production scope;
5. Wuhan Zhengtong Pharmaceutical Co., Ltd. "Drug Production License" production address and production scope of production (trusteter);
6. Hubei Province Hongyuan Pharmaceutical Technology Co., Ltd.'s "Drug Production License" production address and production range and production scope of production (commissioner);
7. Changda Pharmaceutical (China) Co., Ltd.'s "Drug Production License" production address and production scope of production (commissioner);
8. Hubei Kang Weiyuan Pharmaceutical Co., Ltd.'s "Drug Production License" production address and production scope of production (commissioner);
9. Hubei Meisuke Pharmaceutical Co., Ltd. "Drug Production License" production address and production scope of production (commissioner);
10. Wuhan Jestbang Health Products Co., Ltd. "Drug Production License" production address and production scope of production (commissioner);
11. Hubei Ruize Pharmaceutical Co., Ltd.'s "Drug Production License" production address and production scope of production (commissioner);
12. Hubei Minkang Pharmaceutical Group Co., Ltd. "Drug Production License" production address and production scope of production (commissioner);
13. Hubei Xin Ankang Pharmaceutical Co., Ltd.'s "Drug Production License" production address and production scope of production (trusteter);
14. Changesa Life Science (Wuhan) Co., Ltd. Drug production license production scope (including the scope of production scope of text) changes;
15. Sino -US Huasitong Biomedical Technology (Wuhan) Co., Ltd. "Drug Production License" production address and production range and production scope of production (commissioner);
16. Hubei Hengan Fulin Pharmaceutical Co., Ltd. adds new production workshops;
17. Hubei Kang Weiyuan Pharmaceutical Co., Ltd.'s "Drug Production License" production address and production scope of production (commissioner);
18. Wuhan Biological Products Research Institute Co., Ltd. New production workshop production line;
19. Hubei Yuanda Life Science and Technology Co., Ltd. changed its legal representative, person in charge of the enterprise, person in charge of quality, and quality authority;
20. Wuhan Xianlu Pharmaceutical Technology Co., Ltd.'s "Drug Production License" production address and production range of production and production scope (commissioner).
(2) Drug production license issuance and change
1. Jianchi County Traditional Chinese Medicine Hospital changed the registered address and quality leader.
2. Production permit for medical device
The production license of medical device production licenses is issued, continued, and changed
1. The issuance
2. Senmo City Songqing Plastic Products Co., Ltd. issued;
3. Macheng City Binfeng Protection Products Factory issued;
4. The person in charge of the enterprise of Wuhan Youzhiyou Medical Technology Co., Ltd. changed;
5. Wuhan Nanxue Pharmaceutical Co., Ltd. increases production products (including changing production scope);
6. The name of the enterprise, legal representative, and production address of the Wuhan Jindian Yiyi Preparation Center;
7. Microchor (Wuhan) Medical Technology Co., Ltd. has increased production products by the trustee;
8. Jianghe Medical Materials Co., Ltd., Qianjiang City, Hubei Province, increase production products (including changing production scope);
9. Hubei Meishunhe Medical Technology Co., Ltd. increases production products (including changing production scope);
10. Hubei Kangquan Medical Technology Co., Ltd. increases production products (including changing production scope).
3. Drug management license
(1) Drug business license renewal and change
1. Hubei Yongshun Xinxin Pharmaceutical Co., Ltd. replaced;
2. Hubei Jiuzhou Hengjian Medical Co., Ltd. changes the registered address, warehouse address, corporate legal representative, and head of the enterprise;
3. Hubei Qi'an Pharmaceutical Co., Ltd. replaced;
4. The person in charge of the quality of the quality of Hubei Jianjiantang Pharmaceutical Co., Ltd.;
5. Yichang Guoji Pharmaceutical Co., Ltd. changes the name, registered address, and warehouse address of the enterprise;
6. Hefeng County Shengxingxiang Pharmaceutical Chain Co., Ltd. changes quality leaders;
7. Wuhan Minghui Pharmaceutical Development Co., Ltd. changes quality leaders;
8. Huayuan Pharmaceutical (Yichang) Co., Ltd. change quality leaders;
9. Wuhan Meikangyuan Pharmaceutical Co., Ltd. changes the person in charge of the enterprise, the person in charge of quality, and the scope of the nuclear reduction;
10. Chongqing Pharmaceutical Yichang Hengkang Medical Co., Ltd. changes the legal representative of the enterprise;
11. Wuhan Xianhui Pharmaceutical Co., Ltd. changes the warehouse address and business scope;
12. Wuhan Emerging Elite Pharmaceutical Co., Ltd. changes the registered address.
(2) Examination and approval of special drugs
1. The legal person of the National Medicine Gezhouba Central Hospital;
2. Wuhan Ninja Pharmaceutical Co., Ltd. Non -pharmaceutical manufacturers purchase caffeine raw materials for approval;
3. Huangshi Shixing Pharmaceutical Co., Ltd. Pharmaceutical manufacturers use the standard products of anesthesia drugs, psychotropic drugs, and control products to purchase approval;
4. Yongan Health Pharmaceutical (Wuhan) Co., Ltd. non -pharmaceutical manufacturer purchase and approve the purchase of caffeine raw materials.
Fourth, drug and medical device advertising approval
1. 1 piece of drug advertising approval Hubei Dongxin Pharmaceutical Co., Ltd. (article);
2. Drug advertising approval Hubei Hengan Fulin Pharmaceutical Co., Ltd. (text); 3. 3 pieces of drug advertising approval Hubei Kotian Pharmaceutical Co., Ltd. (text);
4. 2 pieces of drug advertising approval Hubei Fucheng Tian Pharmaceutical Co., Ltd. (text);
5. Drug advertising approval Hubei Ye Zhentang Pharmaceutical Co., Ltd. (text);
6. Drug advertising approval of Ma Yinglong Pharmaceutical Group Co., Ltd. (text);
7. 2 pieces of drug advertising approval Wuhan Beishen Pharmaceutical Co., Ltd. (text);
8. 3 pieces of drug advertising approval Wuhan Nuo Pharmaceutical Co., Ltd. (see);
9. 2 pieces of drug advertising approval Wuhan Taifu Pharmaceutical Co., Ltd. (text);
10. Pharmaceutical advertising approval Wuhan Wujing Pharmaceutical Co., Ltd. (article);
11. Pharmaceutical advertising approval of Wuhan Yuanda Pharmaceutical Group Sales Co., Ltd. (see);
12. Medical Device Advertising Approval Ogilvy Medical Products Co., Ltd. (text);
13. Medical Device Advertising Examination and Approval Bo Lianzhong Science (Wuhan) Technology Co., Ltd. (text);
14. Medical Devices Advertising Examination and Approval Hubei Bairun Medical Technology Co., Ltd. (text) 4 pieces;
15. Medical Device Advertising Approval Hubei Enwei Pharmaceutical Co., Ltd. Jingzhou Branch (text);
16. Medical Devices Advertising Examination and Approval Hubei Faipu Rui Protection Technology Co., Ltd. (text);
17. Medical Device Advertising Approval Hubei Full Niu Medical Gel Co., Ltd. (text);
18. 6 pieces of medical device advertising approval Hubei Hongshang Pharmaceutical Co., Ltd. (text);
19. Medical equipment advertising approval Hubei Huizhenyuan Pharmaceutical Co., Ltd. (text) 4 pieces;
20. Medical Device Advertising Touring Hubei Junyan Medical Technology Co., Ltd. (text);
21. Medical Devices Advertising Examination and Approval Hubei Linyuantang Pharmaceutical Co., Ltd. (text);
22. Medical Device Advertising Examination and Approval Hubei Longwei Pharmaceutical Co., Ltd. (text);
23. Medical equipment advertising approval Hubei Meishunhe Medical Technology Co., Ltd. (text);
24. Medical equipment advertising approval Hubei Mulan Tongda Technology Co., Ltd. (text);
25. Medical Device Advertising Examination and Approval Hubei Pu'ai Pharmaceutical Co., Ltd. (text) 3 pieces;
26. Medical equipment advertising approval Hubei Senxin Biotechnology Co., Ltd. (text);
27. Medical Devices Advertising Examination and Approval Hubei Hengry Medical Device Co., Ltd. (text);
28. Medical Devices Advertising Approval of 1 piece (text) of Qianjiang Jianghe Medical Materials Co., Ltd. (text);
29. Medical Devices Advertising Examination and Approval Hubei Yihuan Medical Device Co., Ltd. (text);
30. Medical Devices Advertising Examination and Approval Hubei Yigengtang Technology Co., Ltd. (text);
31. Medical Devices Advertising Examination and Approval Hubei Zhongsen Great Health Co., Ltd. (text) 8 pieces;
32. Medical Devices Advertising Advertising Twenty -two pieces of Jinyu Long Medical Technology (Hubei) Co., Ltd. (text);
33. 10 pieces of medical device advertising approval Kingsda Medical (Xianning) Co., Ltd. (text);
34. Medical Device Advertising Examination and Approval Menrini (China) Investment Co., Ltd. (text);
35. Medical equipment advertising approval Qianyangtian (Wuhan) Industrial Co., Ltd. (text);
36. Medical equipment advertising approval Tianjin Zhongsheng latex Co., Ltd. (text);
37. Medical equipment advertising approval Stable Medical (Wuhan) Co., Ltd. (text) 2 pieces;
38. Medical Devices Advertising Advertising Wuhan Caizhitang Biomedical Co., Ltd. (text) 3 pieces;
39. Medical Devices Advertising Advertising Wuhan Huasida Protective Products Co., Ltd. (text);
40. Medical Device Advertising Advertising Wuhan Huawei Technology Co., Ltd. (text);
41. Medical Device Advertising Advertising Wuhan Health Xintiandi Technology Co., Ltd. (text);
42. Medical equipment advertising approval Wuhan Jestbang Health Products Co., Ltd. (text);
43. Medical Device Advertising Advertising Wuhan Li Jitang Biotechnology Co., Ltd. (text);
44. 1 piece of medical device advertising approval Wuhan Nanxue Pharmaceutical Co., Ltd. (text);
45. Medical Devices Advertising Advertisement and Approval Wuhan Sitari Medical Device Development Co., Ltd. (text);
46. 9 pieces of medical device advertisements Wuhan Yurui Technology Co., Ltd. (text);
47. Medical equipment advertising approval Wuhan noon Sunshine Pharmaceutical Biotechnology Co., Ltd. (text);
48. Medical Devices Advertising Advertising Wuhan Zhiyin Hall Biotechnology Co., Ltd. (text);
49. Medical equipment advertising approval of Xiantao City Daoqi Plastic Industry Co., Ltd. (text);
50. Medical equipment advertising approval Xianning Aiko Medical Products Co., Ltd. (text) 5 pieces;
51. 3 pieces of medical device advertising approval Xiangyang City Yangshiro Biotechnology Co., Ltd. (text).
5. Drug export sales certificate
1. Yichang Renfu Pharmaceutical Co., Ltd.
2. Yichang Renfu Pharmaceutical Co., Ltd.
3. Yichang Renfu Pharmaceutical Co., Ltd. (Midazolun injection 1ml: 5mg);
4. Yichang Renfu Pharmaceutical Co., Ltd.
5. Wuhan Biological Products Research Institute Co., Ltd.;
6. Renfuk Pharmaceutical (Wuhan) Co., Ltd.
6. Internet drug information service qualification certificate
1. Da Ginlin (Hubei) Pharmaceutical Chain Co., Ltd. changes the name of the enterprise, website name, and unit address;
3. The issuance of Wuhan Changshun Pharmacy Co., Ltd.;
4. The issuance of Yuhang Drug Supermarket in Lishui County;
5. The issuance of Wujidang Da Pharmacy Chain Co., Ltd.
6. Wuhan Hao Pharmacist Xiang Song Da Pharmacy Co., Ltd. changes the name, website name, legal representative, unit address;
7. Wuhan Jiaxintang Pharmaceutical Chain Co., Ltd. issued;
8. Hubei Meisuke Pharmaceutical Co., Ltd. issued;
9. The issuance of Shansangtang Da Pharmacy in Shashi District;
10. Wuhan Guantong Medical Co., Ltd. issued;
11. Hubei Jianyu Technology Co., Ltd. issued;
12. Substantial Daida Medical Technology (Wuhan) Co., Ltd. was canceled.
Seven, cosmetics production
1. The person in charge of quality and safety of Odima (Hubei) Biotechnology Co., Ltd.;
2. Hubei Aiyuan Ai Ai Technology Co., Ltd. continues.
Eight, epidemic prevention and control medical device production enterprise approval
1. Xiantao Songqing Plastic Products Co., Ltd. issued a medical surgical mask and used medical masks at one time;
2. Macheng City Binfeng Protection Products Factory issued a one -time use of medical masks.
(Source: Hubei Provincial Drug Administration)
【Edit: Wang Yujin】
- END -
Spleen deficiency is equivalent to half of it?People with spleen deficiency often have 6 kinds of performance, you should raise the spleen

When it comes to deficiency, many people are the first association of kidney defic...
Women exercise violently after the injury, the small bag becomes a big hematoma on the fist
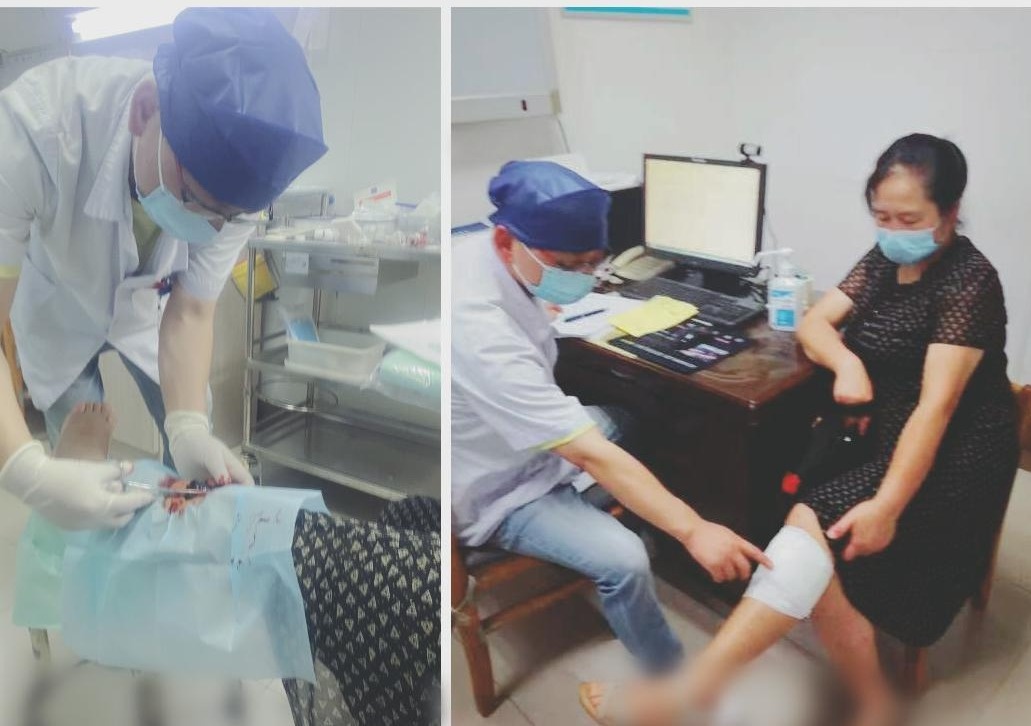
The Yangtze River Daily Da Wuhan Client June 29th (Correspondent Zhou Shan) On Jun...