Inventory 7 classic antiviral drugs approved by the FDA in the United States
Author:Yaizhi.com Time:2022.09.26
Inventory 7 classic antiviral drugs approved by the FDA in the United States
Source: Bo Ya/Qingmei
For many years, researchers have been developing effective antiviral therapy. Although many early attempts were invalid, even took some detours.
VAN ROOYEN and Rhodes proposed the concept of special treatment for some virus infections in "Human Viral Diseases" published in 1946. The focus is on the use of antibiotics, including sulfa drugs, for the treatment of epidemic mumps and ceilings. But now we know that antibiotics for virus infections are usually invalid.
In 1957, Esther and Linden Manumon described the famous interferon for the first time. Subsequently, human interferon was developed to treat specific virus infections, namely hepatitis B and the nearest hepatitis C virus infection.
However, interferon found in the late 1950s may also be another detours of antiviral therapy.
Time has come to the end of the 1950s. With 5-iodine -2′-deoxyladin, that is, iodide, was synthesized, and showed excellent antiviral activity, the development of antiviral drugs was Entering a high -speed development period in ten years.
From 1960s to the present, about 90 antiviral drugs have been approved. Recently, ACS C & EN has invested 7 milestones in the development of antiviral drugs in the past few decades.
Picture source: ACS C & EN
1. 1963: Idoxuridine -the first useful antiviral nucleoside analog
Picture source: reference literature
In the field of antiviral, many people think that Dr. Bill Prussov described the 5-iodine -2′-deoxyladine (IDU) and realizing its antiviral characteristics in 1959 is the most important early milestone.
However, this nucleoside analog and similar nucleoside analogs were first published in cancer magazines. The purpose is to develop molecules that interfere with DNA synthesis in order to produce cells inhibitors or cytotoxic drugs for treating tumor diseases. However, the unexpected harvest of this work is to find that IDU is a specific inhibitor of certain large DNA viruses, and the most obvious is herpes simplex virus (HSV).
This compound is still in use today, with cytotoxicity, so it is only applicable to local applications.
2. 1977: Arabinoside -the first antiviral compound that can be administered in the whole body
Picture source: reference literature
In 1964, M. Privat de Garilhe and J. De Rudder first described the antiviral activity of Ara-A) of Ara-A for the first time.
ARA-A is the first compound that has sufficient non-toxicity in nucleoside analogs that can be administered throughout the body. Dr. Richard Whitley proves the clinical value of this compound. For the first time, the study shows that as long as the disease starts in the early stage of the disease, it is possible to curb the straw of the immunosteriator, reverse the herpes encephalitis and the newborn occasionally happened occasionally Potential fatal progress of overwhelming herpes infection.
3. 1981: ACYCLOVIR -the first high -selective antiviral compound
Picture source: reference literature
Agello has a huge influence in the field of antiviral drugs and is the first high -selective antiviral compound. It is called the prototype of the "second -generation" nucleoside analogy, which is listed in the UK as a non -prescription drug.
Agelovovir is a substrate for deoxidine kinase encoded by HSV, which is usually called thymosine kinase (TK). It is an effective suicide inhibitor of herpes specific DNA polymerase.
Agelloville has been recognized as one of the safest drugs. In the process of decades of use, almost no adverse reactions have occurred (except for the low -water solubility of the compound), including those who have used the compound for more than 20 years to have been used for more than 20 years to use the compound for more than 20 years. Patients who suppress recurrence HSV.
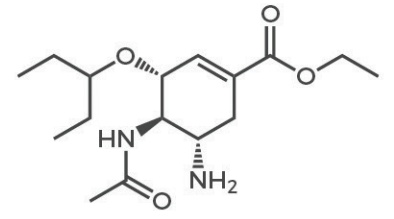
4. 1987: Azidothymidine -the first effective anti -HIV virus drug
Picture source: reference literature
In the early 1980s, when HIV/AIDS appeared as a major new viral disease, the science of antiviral research was already very developed. The first effective antiviral compound (AZT, Zidov) has been screened in the compound library screened by Burrows Wellcom and the National Cancer Institute (USA), and was quickly reported as a reverse virus (including HIV) in 1985 Specific inhibitors.
The action mechanism of AZT is based on the drug -based phosphoric acid of the drug into AZT trigensive, and then interacts with the binding site of the substrate of the HIV reversal recording enzyme, thereby playing the role of a chain termination agent. After discovering the AZT, several other types of bispernamucleoside (DDI) analogs (DDI, DDC, D4T, 3TC, ABC, (-) FTC) were found. The formal permidase inhibitor) is formally licensed for HIV infection. All these NRTISs are similar: After they are phosphorylated into triple phosphate, they are interacted as the "chain terminal" of HIV reversal recording enzymes, thereby preventing the formation of the primary virus DNA, otherwise the infection state will eternity (in the original original (in the original original (in the original original (in the original original (in the original original (in the original (in the original (in the original (in the original (in the original (in the original (in the original (in the original (in the original (in the original (in the original, The virus DNA is integrated after the host cell genome). 5. 1995: Saquinavir -The first antiviral drug for HIV virus protein
Picture source: reference literature
The interpretation of the HIV genome has revealed that the HIV virus has a specific protease, which provides targets for specific antiviral drug development.
Roche chemist and molecular virusist team led by Dr. Joe Martin and Dr. Roberts, Dr. Norol, and Dr. Roberts, achieved the synthesis of Shaquinaov, the first peptide-based transition mixture. Shaquinaov is proved to be active under the concentration of Nonmore and is one of the most powerful antiviral substances described so far. Soon, Shaquanov was replaced by similar compounds produced by several other competitive companies.
At present, seven protease inhibitors are authorized to treat HIV infections: Sakanova, Lito Navavvi, Nikonova, Andinavi, Ripanova and Azamova Essence
6. 1999: OSELTAMIVIR -
Picture source: reference literature
M. Von Itzstein and his colleagues designed several salivary acid analogs Neu5ac2en according to the crystal structure of the influenza neuropine (NA) in 1993, and developed 4-胍 base -Neu5ac2en (Relenza). This is The first NA inhibitor was sold by Glaxo.
However, the oral use of this compound is poor, so it must be applied by inhalation. Subsequently, other NA inhibitors were developed, including more successful Olydivir (developed by Gilead Sciences, C.U. Kim et al. First reported for the first time in 1997). Osdowei introduced the cycloonne ring in the structure and replaced the polar glycerin with a more lipid side chain. Osdowe is a pre -ethyl medicine, which has oral biological utilization and is easily converted into active carboxylic acids in the liver.
At present, Osdowe is still used for the treatment of anti -influenza treatment.
7. 2020: RemDesivir -Extensive anti -RNA virus activity
Picture source: reference literature
Redsovir (GS-5734) is a nucleotide analogy medicine that disturbs virus copying. It was initially evaluated as a burst of Ebola virus in clinical trials in 2014. With the confirmation of the extensive anti -RNA virus activity of Rydessoviri, multiple groups have evaluated the antiviral activity of in vitro and in vivo, and verified its activity to coronary virus, including SARS and MERS. With the outbreak of COVID-19, Redcie was approved by the FDA for the treatment of the new crown in 2020.
Troop
In China, the approved antiviral drug indications are concentrated in virus infections such as hepatitis B, hepatitis C, flu, mumps, and mumps. The research indications are also concentrated in virus infections with high incidence of hepatitis B, hepatitis C, and influenza virus.
In the field of antiviral infection, key enterprises include Guangdong East Sunshine Pharmaceutical, Shanghai Biological Products Research Institute, Zhengda Tianqing, Fujian Guangshengtang, etc.
Key enterprises in the field of Chinese antiviral drugs are under research on projects
Data source: medicine wisdom data
After decades of antibiotics, antiviral drugs have finally ushered in their own era. At present, dozens of antiviral drugs have been formally approved to treat viral diseases. Their application is mainly to treat herpes virus (HSV, VZV, CMV) and HIV, HBV, HCV, and flu virus infections.
At present, the genome sequences of many viruses can be obtained, and the structure and function of the virus protein have also been known, so as to determine the new specific goals for reasonable drug design, but the difficulty of transforming specific inhibitors into effective drugs is still a pharmaceutical chemist The main task.
Disclaimer: This article is the content of Yaozhi.com, and the copyright of the pictures and text belongs to the original author. The purpose of reprinting is to pass more information, which does not represent the viewpoint of this platform. If the content, copyright and other issues are involved in the work, please leave a message on this platform, and we will delete it as soon as possible.
- END -
Low the risk of epidemic in some areas of Jining City

Qilu.com · Lightning News, September 12th, on September 12, the Jining Municipal ...
From 0:00 on September 20, 2022 to 24:00, there are no new local diagnosis cases, and 1 case of new native symptoms.
From 00:00 on September 20, 2022, there were no new local diagnosis cases in the city, and one of the new native symptoms was added.From 0:00 on September 20th to 24:00, there were no new medical obse